|
Nuclides that have the same neutron number but a different proton number are called isotones. Neutron number is rarely written explicitly in nuclide symbol notation, but appears as a subscript to the right of the element symbol. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. The nucleus is composed of protons and neutrons. While this is not energetically unbalanced, it requires that an electron get very close to the nucleus, which is extremely rare under the rules of quantum mechanics.The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. 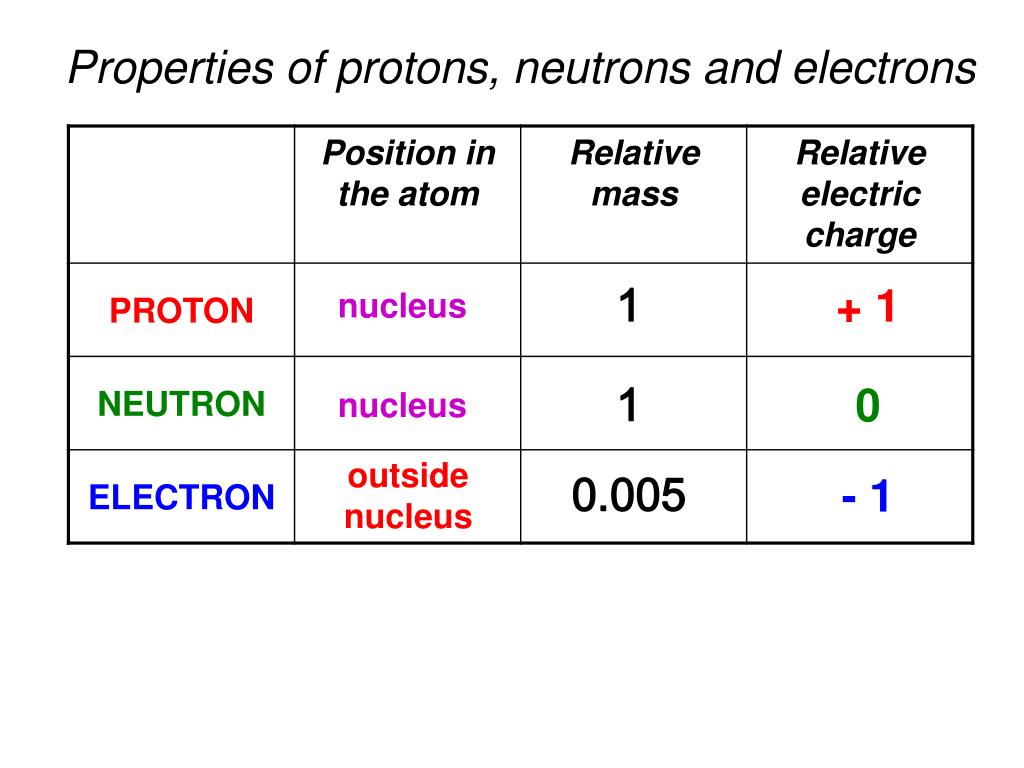
Another is the "electron capture" event, in which a proton captures one of the electrons circling the nucleus, turns into a neutron, and emits an electron neutrino. This action, by itself, is forbidden by conservation of energy, so it can only occur in nuclei that have unusual energy configurations. One is the "beta-plus" decay, in which a proton turns into a neutron, emitting a positron (antielectron) and an electron neutrino. There are other, much rarer, radioactive decays involving the weak interaction. That boson quickly decays into an electron and an antineutrino. During beta decay, one of the "down" quarks turns into a "up" quark, with the extremely short-lived "weak boson" W- carrying off the charge difference of -1. The "up" quark has an electric charge of +2/3, while the "down" quark has a charge of -1/3. The proton consists of two "up" quarks and one "down" quark. The neutron consists of an "up" quark and two "down" quarks. Under the quark model, the transmutation of a neutron into a proton is actually a transmutation of one of its constituent quarks. It took the development of quantum mechanics, and the discovery of the neutron, to realize that the former model was incorrect. Since the neutron and proton have nearly equal mass, and the mass of the electron is negligible in comparison, either model was consistent with observed nuclear charges and atomic weights. The resulting Calcium-40 nucleus has 20 protons and 20 neutrons. When it undergoes beta decay, one of the neutrons turns into a proton, an electron, and an antineutrino. We now know that Potassium-40 has 19 protons and 21 neutrons. When it underwent beta decay, one of the nuclear electrons escaped, resulting in a Calcium-40 nucleus with 40 protons and 20 electrons, and a net positive nuclear charge of 20. This gave it a net positive nuclear charge of 19. That is, it was believed that, for example, Potassium-40 had 40 protons and 21 electrons in its nucleus, in addition to the 19 electrons circling outside of the nucleus. 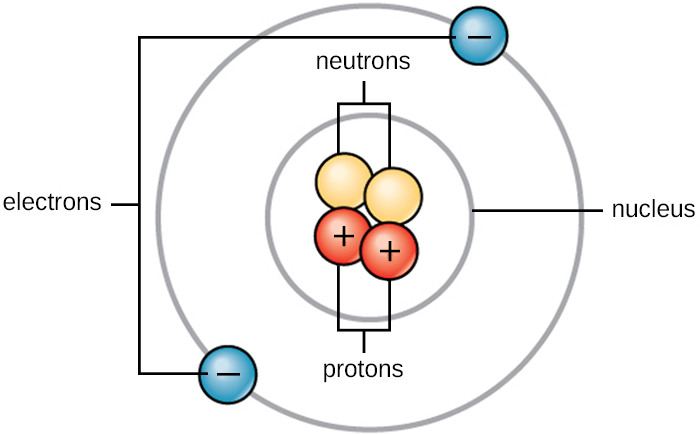
At this time, it was believed that the electrons resided in the nucleus all along, and somehow escaped. The beta particles were later determined to be just electrons. 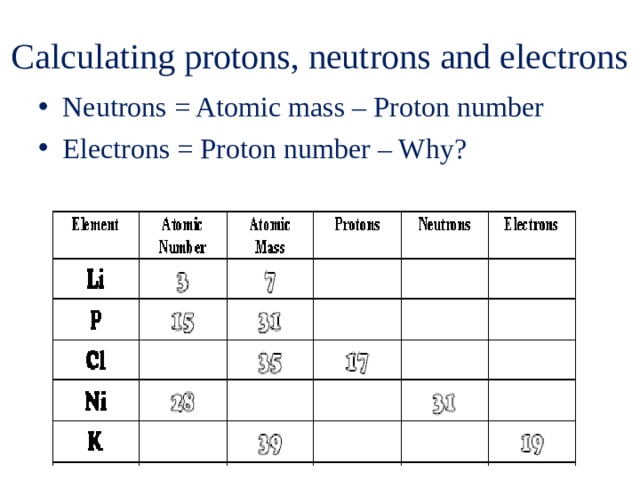
īefore the structure of the nucleus was understood, this emission of negative particles was observed, and they were called "beta particles" or "beta rays". The most common radioactive decay involving the weak interaction is the transmutation of a neutron into a proton, with emission of an electron and an antineutrino. It is one of three major types of radioactivity (the other two being alpha decay and gamma decay), and the only one that involves the transmutation of a subatomic particle. The Feynman diagram for the beta decay of a neutron into a proton, electron, and electron antineutrino via an intermediate heavy W-bosonīeta decay is a type of radioactive decay involving the "weak force" and the transmutation of nucleons (ultimately, the transmutation of quarks) inside an atomic nucleus. Subject classification: this is a science resource.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
